Travere Therapeutics Still On The Rise After Encouraging Data Travere Therapeutics (NASDAQ: TVTX) is up a whopping 75.81% in the past three months, as shares of the biotech company climb out of a steep correction.
By Kate Stalter •

This story originally appeared on MarketBeat

is up a whopping 75.81% in the past three months, as shares of the biotech company climb out of a steep correction.
Biotech companies are almost inherently volatile. So much of the path to profitability depends on treatments delivering promising results in clinical trials. Often, these companies are acquired by bigger pharmaceuticals, or they generate revenue by licensing their therapies to larger firms.
San Diego-based Travere Therapeutics is a biopharmaceutical company that develops and commercializes therapies for rare diseases.
The company is currently developing a product called sparsentan, a treatment for rare kidney conditions that often become very serious and lead to late-stage illness.
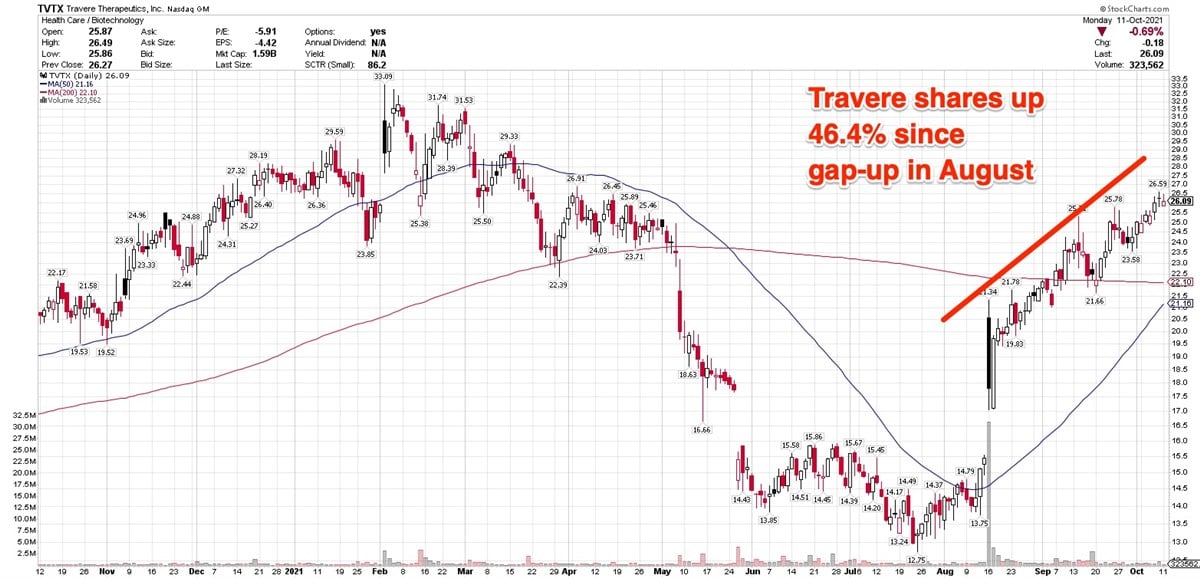
The stock began climbing out of a hole in late July and gapped up 15% in mid-August after the company reported significant reduction in proteinuria, or excessive urine protein.
Patients receiving a regimen of Sparsentan saw a reduction of these protein leaks after 36 weeks.
Investor Optimism In August
That data contributed to the stock's 58.76% rise in August, which occurred in volume that was 139% higher than average. Investors were clearly cheered by the return of Sparsentan as a potential contributor to Travere's growth, after skidding more than 38% in May.
So what caused the May plunge, which is largely responsible for the stock's year-to-date decline of 4.27 %?
The May plummet was due to the FDA's rejection of interim data produced by Travere's Phase 3 Duplex study of Sparsentan in patients with focal segmental glomerulosclerosis (FSGS), a rare and serious kidney disease. Travere has initially released the results on February 2. On that day, the stock gapped up 11% in seven times normal turnover.
Travere had hoped the trial data at 36 weeks would be convincing enough for regulators to fast-track approval of sparsentan.
Since the gap-up in August, Travere shares are up 46.4%, closing Monday at $26.09. For the month of September, the stock advanced 11.09%.
Still A Speculative Stock
So what's next for this stock?
This is potentially a "hopium" play, but that's not uncommon with biotech as investors await a home run. The stock is back to where it was before the May decline, so that's an encouraging sitn that could bode well for further gains.
Travere is a stock I would classify as speculative at this juncture. Despite the undeniably positive trial results, there's still reason to be cautious.
It's certainly possible that the stock could finish the year significantly higher, and investors who believe it's setting up for big gains must realize the risk they are taking.
The company reports its third-quarter on October 28. Analysts expect a loss of $0.72 per share on revenue or $51.30 million.
Travere's revenue currently comes from sales of two product lines.
The first, Thiola, is a medication to control cystine precipitation and excretion in the disease cystinuria, a rare condition characterized by recurrent stones that are difficult to manage.
Current Products Not Blockbusters
Because cystinuria is such a rare condition, Thiola could be considered an "orphan drug," and certainly not one ever destined to be a blockbuster.
The second revenue-producing drug is categorized as bile acid products, including Cholbam. This is a treatment for infants, children and adults with a rare condition called bile acid synthesis disorders.
Adding to the risk of this stock is its small market cap of only $1.58 billion.
In addition to the Duplex study, Travere is also running another, called Protect. This one focuses on use of Sparsentan for treatment of immunoglobulin A nephropathy (IgAN), a rare kidney disorder characterized by buildup of immunoglobulin A (IgA), a protein that helps the body fight infections in the kidneys.
All of this could be very promising, but caution is in order. If FDA approvals come next year, it's possible product launches could begin in 2023.
At this juncture, Wall Street isn't expecting profitability any time soon, but that's exactly what you would expect at this point in the life of a young biotech company.